Designing Covered Cardiovascular Devices? Don’t Forget the Tissue–Cover Interface
- Mar 28
- 3 min read
Teams building covered stents, shunts, flow diverters, LAA devices, heart pumps, or embolic protection systems already juggle materials, mechanics, deliverability, regulatory strategy, and funding. In that complexity, early biological considerations, especially at the interface between tissue and the device covering, often get less attention.
That interface, however, is not a minor detail. It is a dynamic, microscopic environment where blood, vessel or atrial tissue, and the implanted membrane interact continuously.
Addressing it early doesn’t slow development; it can meaningfully reduce downstream risk on the path to first-in-human and beyond.
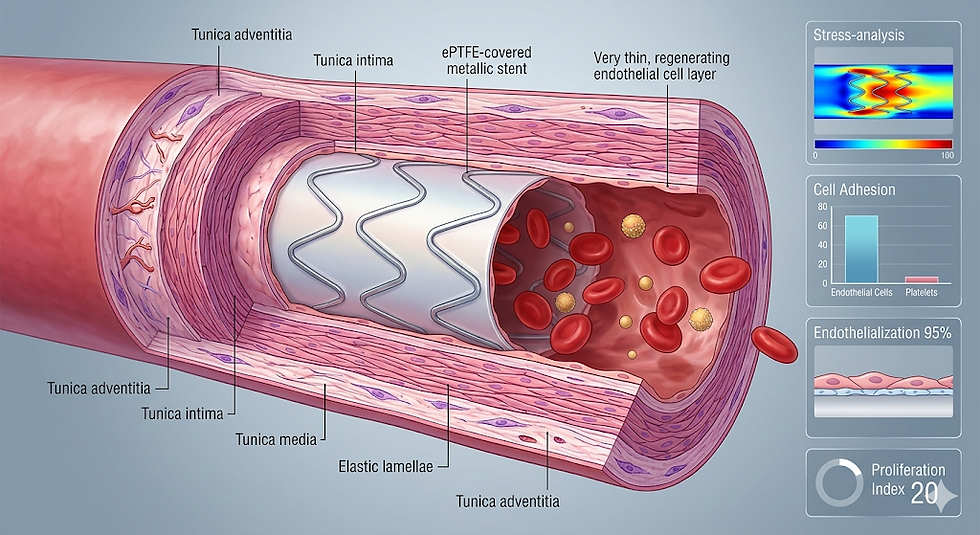
What the tissue–cover interface really is ?
Once a blood-contacting membrane is implanted, it undergoes a fairly consistent biological sequence:
Plasma proteins rapidly adsorb, forming a conditioning layer.
Platelets and coagulation pathways engage with that layer, sometimes triggering thrombus formation.
Endothelial and smooth muscle cells migrate and proliferate, forming neointima.
Macrophages and immune cells influence long-term remodeling and foreign body response.
Clinically, these processes manifest as:
Thrombosis on device surfaces or within covered segments.
Edge restenosis and localized neointimal hyperplasia.
Delayed or incomplete endothelialization.
Excessive tissue ingrowth compromising lumen or flow channels.
Across stents, flow diverters, and related implants, evidence consistently shows that incomplete or heterogeneous endothelial coverage strongly predicts late thrombosis and restenosis.

Where it matters most ?
For many emerging cardiovascular devices, the membrane is not just structural—it actively shapes healing:
Interatrial shunts and structural heart implants.
Temporary or mid-term cardiac assist devices.
Peripheral covered stents (arterial and venous).
Embolic protection filters and occlusion systems.
LAA closure devices.
Flow diverters and flow-modulating implants.
For example, covered stents often show uneven neointimal growth, especially at transition zones.
Flow diverters rely on continuous endothelial bridging across the aneurysm neck; incomplete coverage is linked to delayed occlusion and thrombotic risk.
In both coronary and peripheral applications, immature endothelialization remains a leading predictor of late device failure.
Practical tools you can use
Practical tools you can actually use
You don’t need a full vascular biology program to get meaningful insight. A focused, well‑structured toolkit is often enough.
Hemocompatibility and blood‑flow tests firstThe first tests are typically blood‑flow and hemocompatibility evaluations, grounded in standards such as ISO 10993‑4, which cover endpoints like hemolysis, platelet activation, coagulation, and complement activation. Used thoughtfully, these flow‑based tests can help you:
Discriminate between membrane formulations early in development.
Identify designs that remain stable and safe under adverse or disturbed flow conditions.
Generate data that align cleanly with regulatory expectations and investor due‑diligence needs.
Lean but informative cell studiesOnce blood‑flow behavior looks acceptable, a small, targeted panel of cell assays can be highly revealing:
Endothelial cell adhesion and migration measured directly on actual device or membrane surfaces.
Smooth muscle cell behavior evaluated near edges and step changes that mimic real device geometries.
Sequential blood–cell exposure models, where surfaces are first conditioned with blood and then seeded with vascular cells to better emulate healing.
Programs that drive faster, more complete, and more uniform endothelial and smooth muscle coverage generally align with better biological healing and a lower risk of late thrombosis.
Targeted in vivo correlationFinally, coupling imaging with histology gives disproportionate insight:
OCT or IVUS can quantify apparent surface coverage and neointimal thickness over time.
Histology can then distinguish true endothelialization from organized thrombus and characterize the tissue response.
Flow‑diverter work shows endothelial growth advancing from native vessel segments into treated regions, providing a dynamic picture of healing.
Even relatively small animal studies, if designed with serial imaging and clear biological endpoints, can deliver actionable feedback to guide membrane and device design.
How to integrate this without slowing down ?
A pragmatic development path:
Concept / seed stage
Use ISO 10993-4 as a roadmap.
Run simple flow-loop and endothelial adhesion screens to down-select materials.
Pre–first-in-human
Add platelet activation under relevant shear and basic smooth muscle assays.
Design a focused animal study with clear interface endpoints.
Early clinical
Incorporate advanced imaging where feasible (e.g., OCT).
Align imaging findings with earlier histological insights.
This approach focuses resources on the biological factors most likely to impact trial outcomes, rather than expanding scope unnecessarily.
Why this matters to investors and partners
Programs that demonstrate:
A clear understanding of thrombosis and restenosis mechanisms,
Evidence of faster and more homogeneous healing,
Alignment with recognized standards like ISO 10993-4
are better positioned for regulatory discussions and pivotal trials.
For strategic partners, mechanistic insight into the tissue–cover interface reduces technical uncertainty and accelerates diligence.
References
ISO 10993-4: Biological evaluation of medical devices—Interactions with blood.
Kolandaivelu K. et al. “Stent thrombosis and restenosis: role of endothelial coverage.” PMC (NIH).
Nature Biomedical Engineering and related studies on pro-healing and ultrathin strut stents.
AHA Journals: Flow diverters and endothelialization in aneurysm treatment.
IMR Press (RCM 24.7): Neointimal response in covered stents.
Frontiers in Bioengineering and Biotechnology: Endothelial and smooth muscle cell response to biomaterials.
Marquette University e-publications: Blood–material interaction and endothelialization models.
NAMSA / Measurlabs guidance on hemocompatibility and ISO 10993-4 implementation.





Comments